Walnut Shell Filter Explanation
This page depicts the micro-level mechanism by which black walnut shell filters capture and coalesce oil droplets as part of the de-oiling process in oil production facilities.
There are three parts to this discussion.
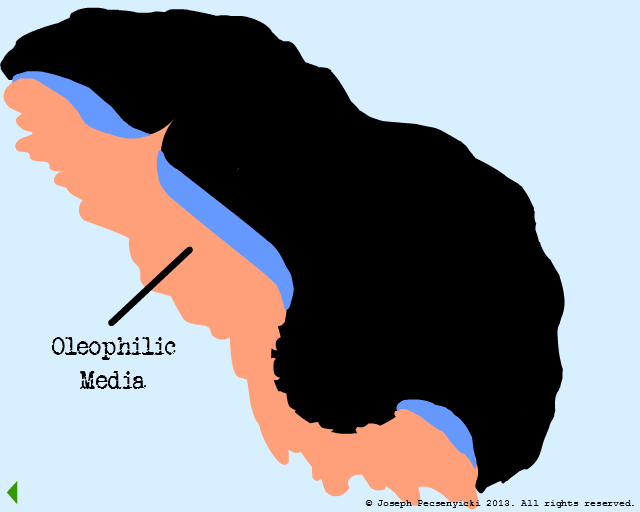
Surface 1 is hypothetical: a completely flat oleophilic (oil-loving) surface.
Surface 2 is also hypothetical: a completely flat oleophobic (oil-hating) surface.
Surface 3 is the actual mechanism for how walnut shell filters work. It features a rough surface with both oleophilic and oleophobic regions, and incorporates aspects of both Surface 1 & Surface 2.
Update
Mar 23, 2021: An animated version of this page was originally created in Adobe Flash in November of 2013. This page covers most of the main points of the animation (using static images published from Flash).
Since Flash was phased out at the end of 2020, the original SWF file can no longer be accessed. The following link is kept for archival purposes only.
Click for the Walnut Shell Filter Flash Animation
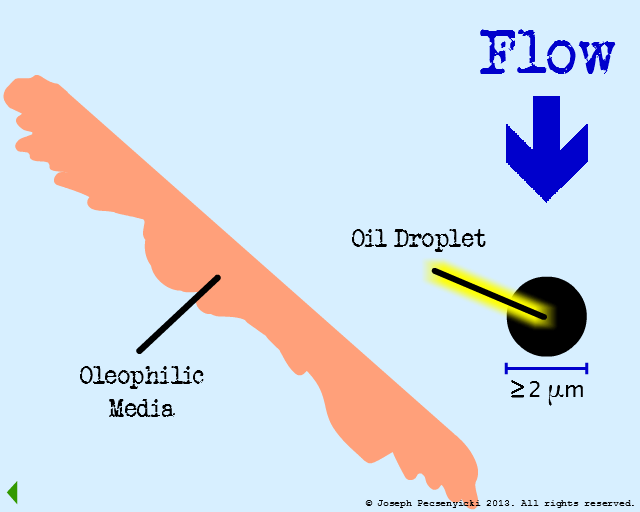
Model #1:
Flat Oleophilic Surface
I
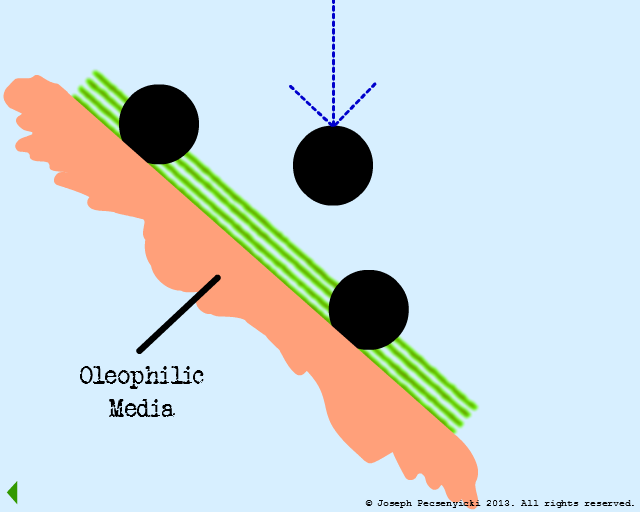
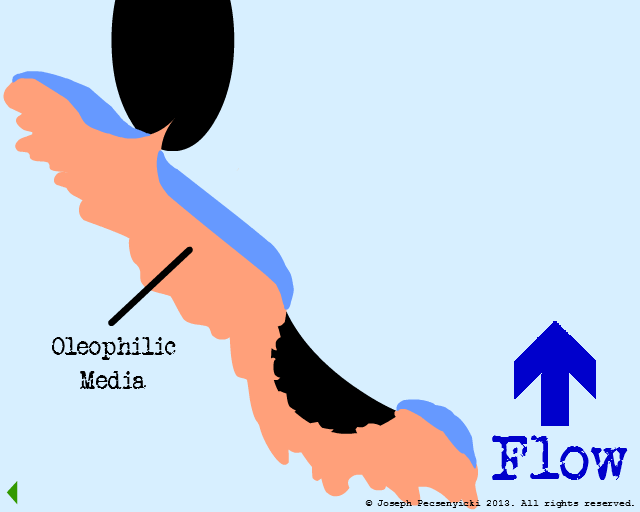
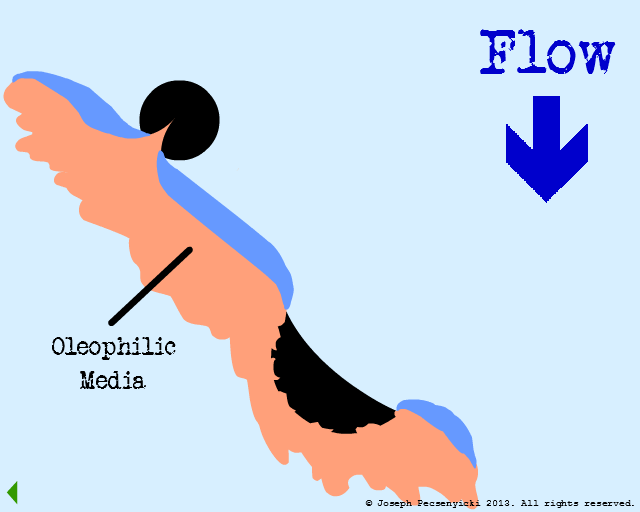
The influent flows through the filter from top to bottom. Best capture rates occur when oil droplet diameter is equal to or greater than 2 microns.
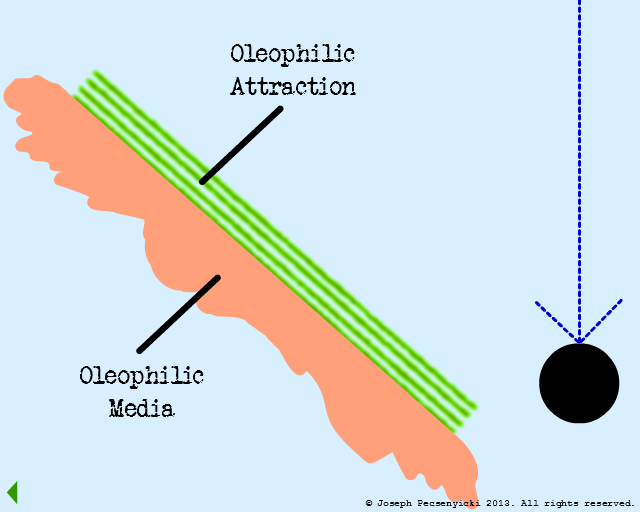
II
Flow through the filter has begun.
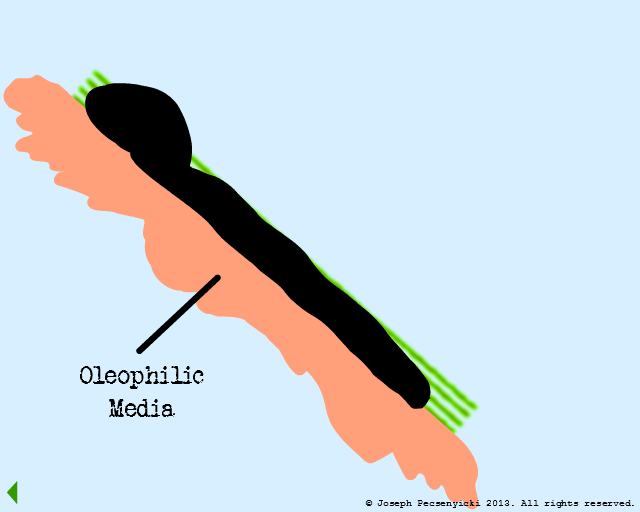
The green area depicts the area where forces of oleophilic attraction occur.
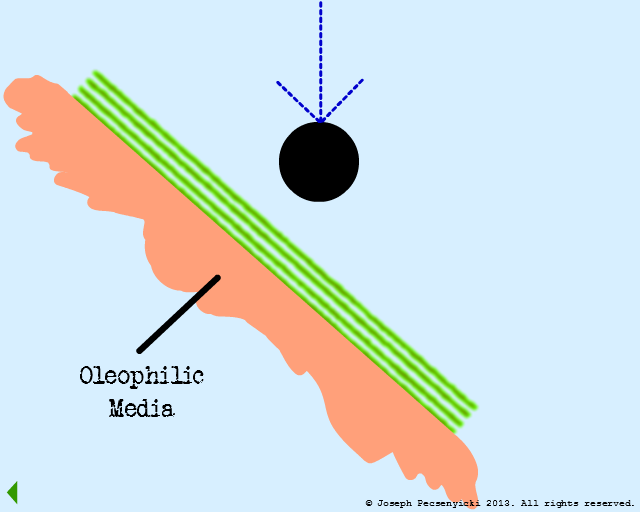
III
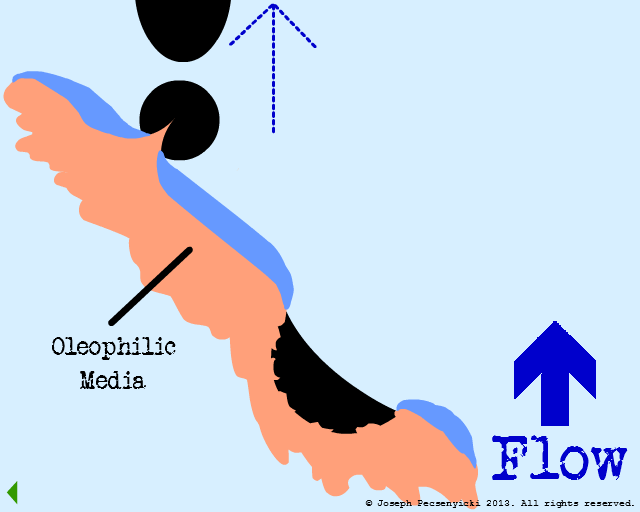
An oil droplet falls & is captured by oleophilic forces on the surface.
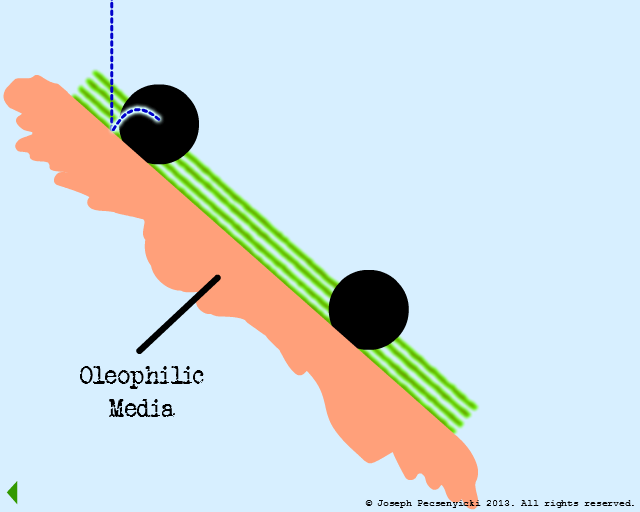
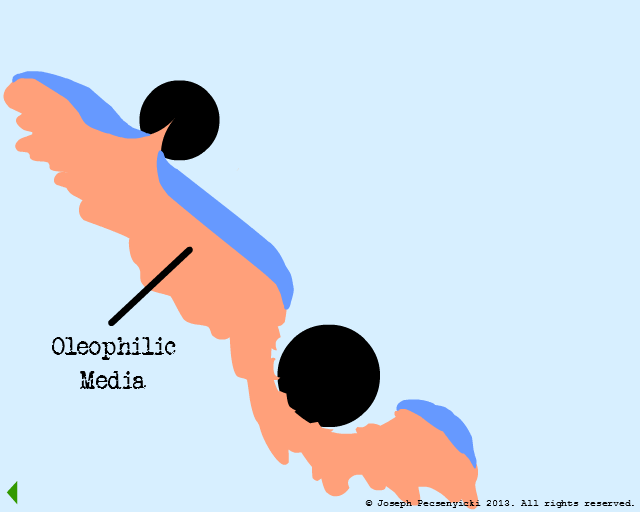
IV
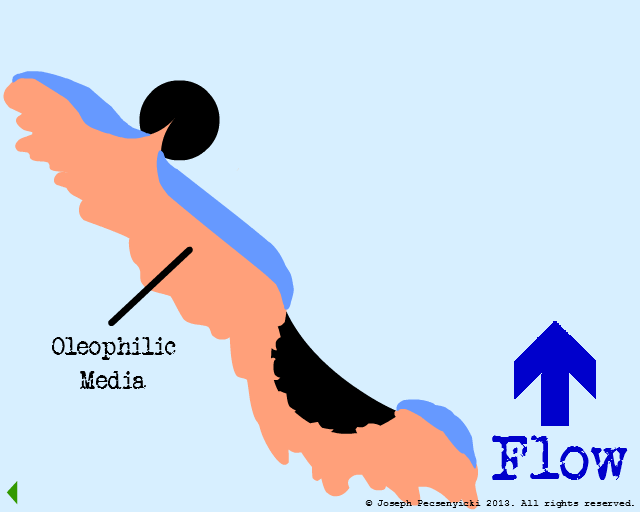
A second oil droplet falls & is also captured on the surface.
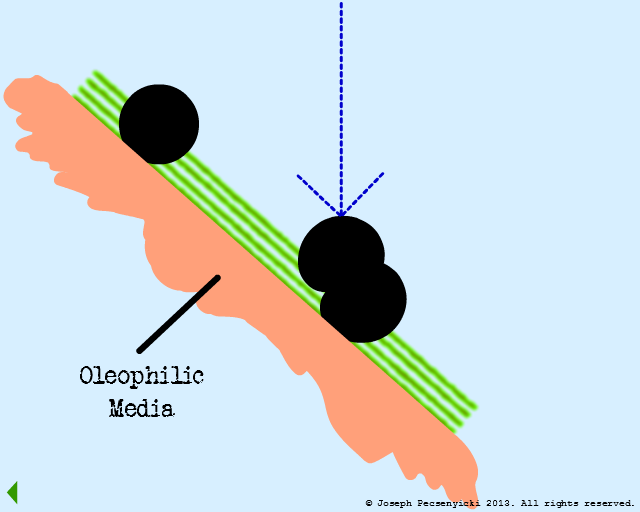
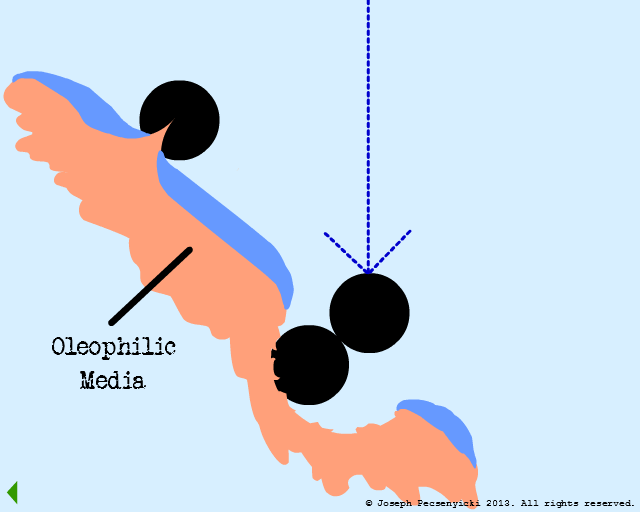
V
A third oil droplet falls to the surface. But this time, it coalesces with one of the previously captured droplets.
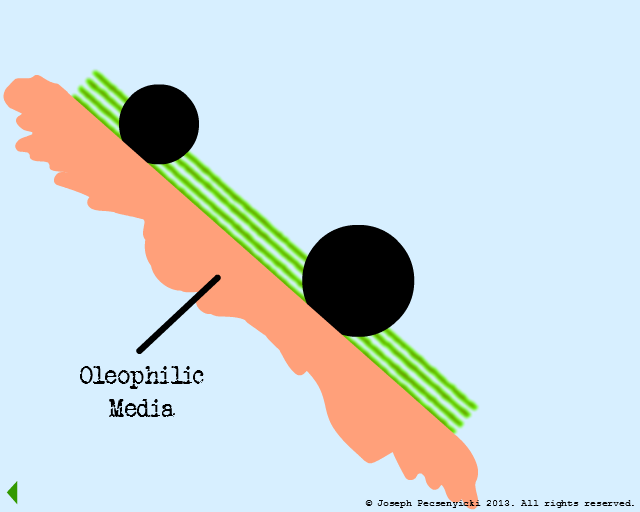
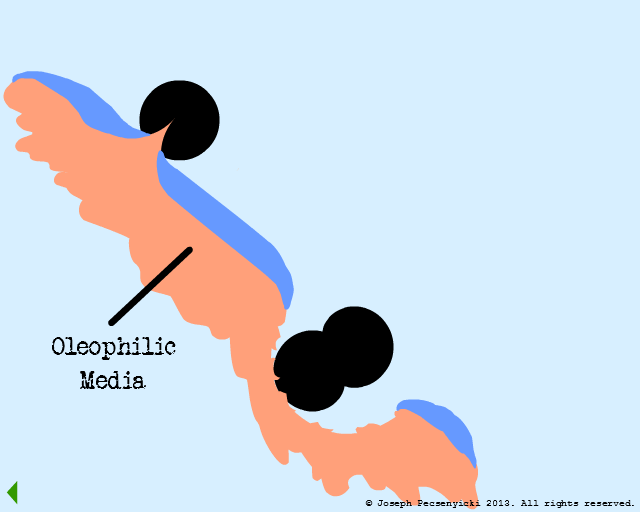
VI
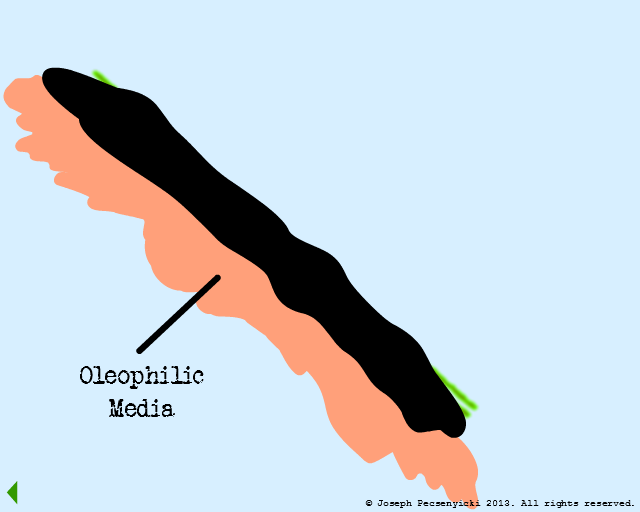
The oleophilic forces help break the surface tension of the large, coalesced oil droplet. The oil begins to cover the filter media surface.
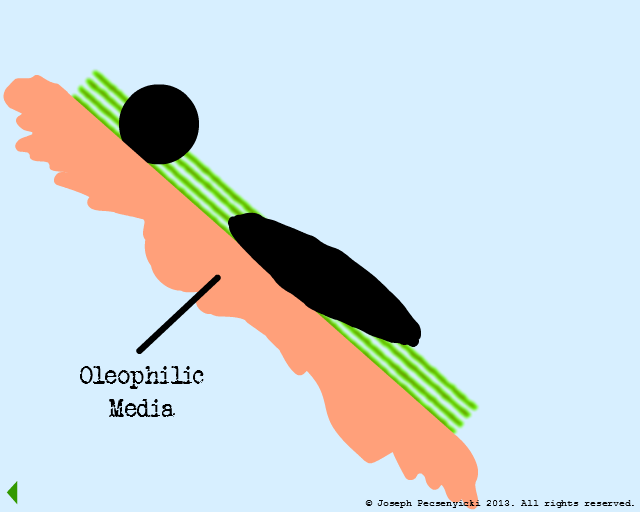
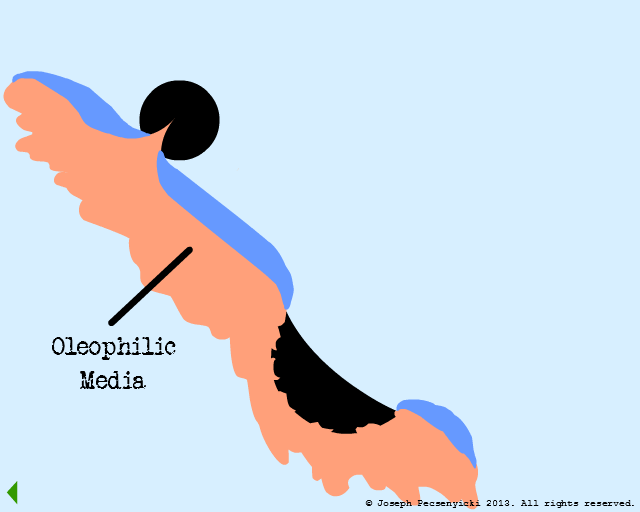
VII
The spreading oil makes contact with another captured droplet, and breaks the surface tension on that droplet as well.
The oil proceeds to wet the entire surface of the oleophilic media.
VIII
The entire surface is completely wetted with oil.
IX
Polyvinyl chloride (PVC) is a good example of this kind of media. It captures oil very well, but gets wetted with oil very easily.
This fouls the media, requiring ever-higher pressures to run influent through the filter.
(And because of its strong affinity for oil, this media cannot be cleaned without using costly chemicals).
Model #2:
Flat Oleophobic Surface
I
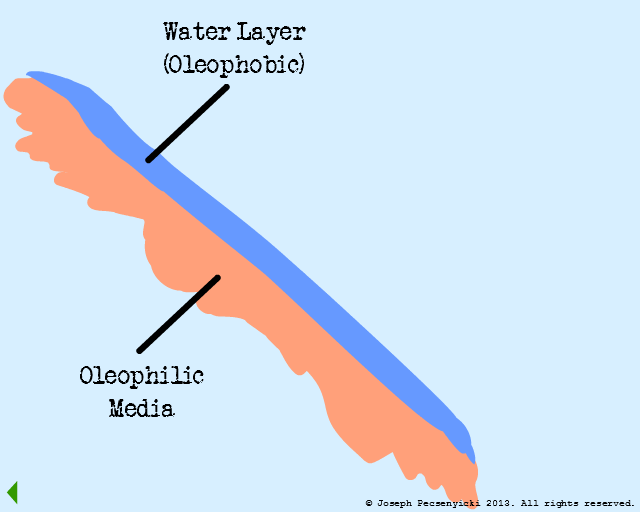
In this model, the flat oleophilic media has been wetted with water for a half-hour.
It is therefore covered with a film of oil-repelling water.
II
Although the media itself is oleophilic, the film of water atop it is oleophobic. Therefore, the latter creates a region of oleophobic repulsion.
III
This type of media never attracts oil droplets, and is of no use whatsoever as an oil filter.
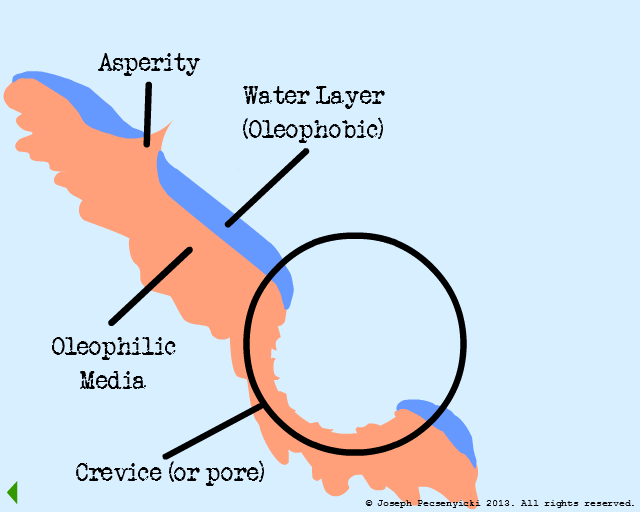
Model #3:
Rough Surface
(Oleophilic & Oleophobic)
I
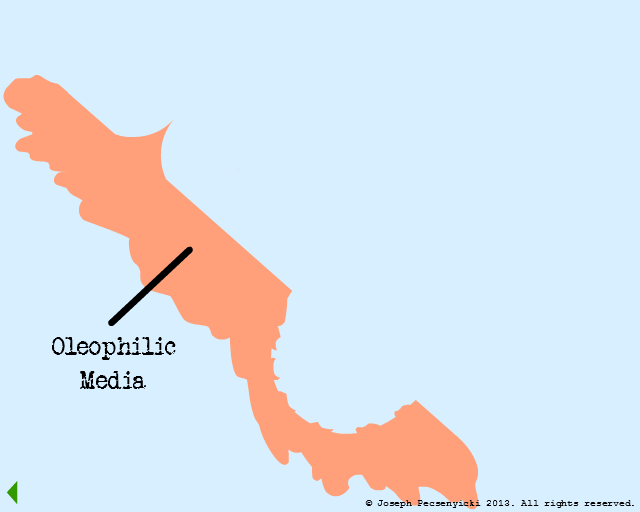
Black walnut shells contain a small amount of natural oils, and are slightly oleophilic.
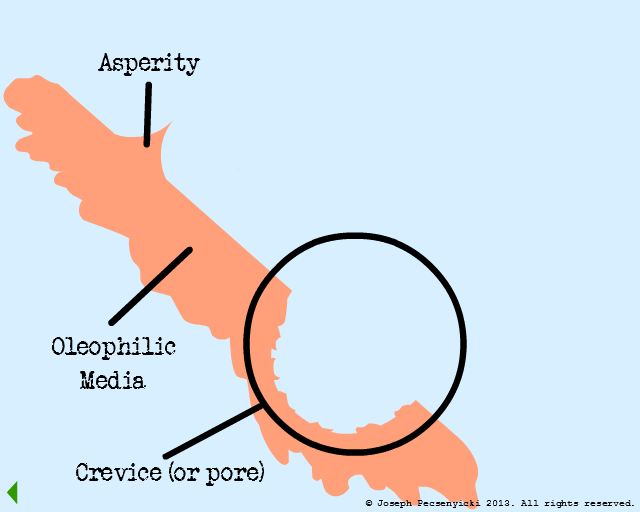
II
Black walnut shells are also rough: they have sharp points (asperities) as well as crevices & pores.
III
The filter is wetted with water for a half hour. This covers the flat surfaces with a layer of oil-repelling water (as in Model #2).
However, the asperities and crevices can still attract oil, since the water layer doesn't completely cover them (as per Model #1).
Apart from any intermolecular forces, oil droplets can also get physically trapped in the crevices (just as dust gets trapped in nooks and crannies in one's home).
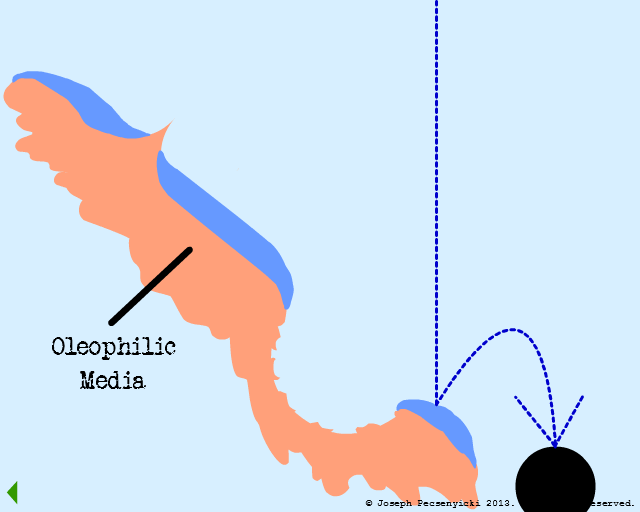
IV
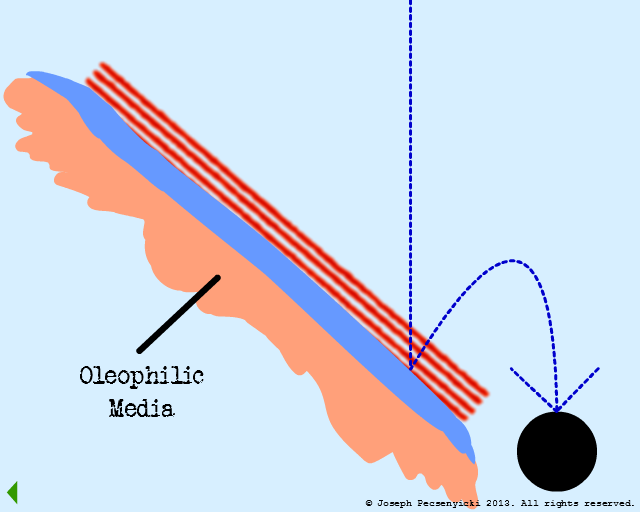
Oil droplets that impact the oleophobic water film are not captured (as in Model #2, Part III).
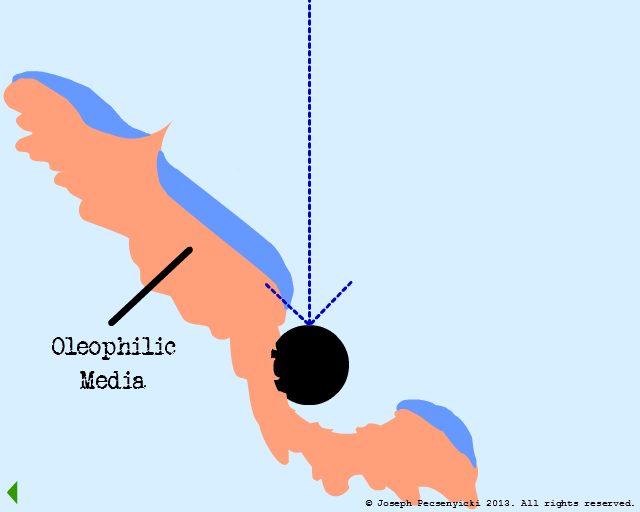
V
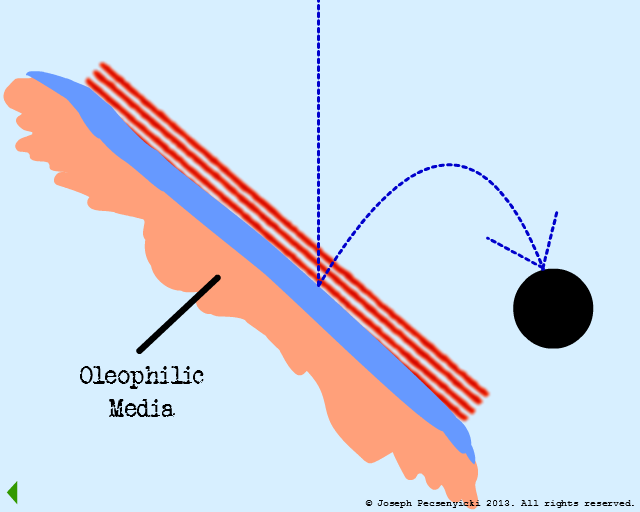
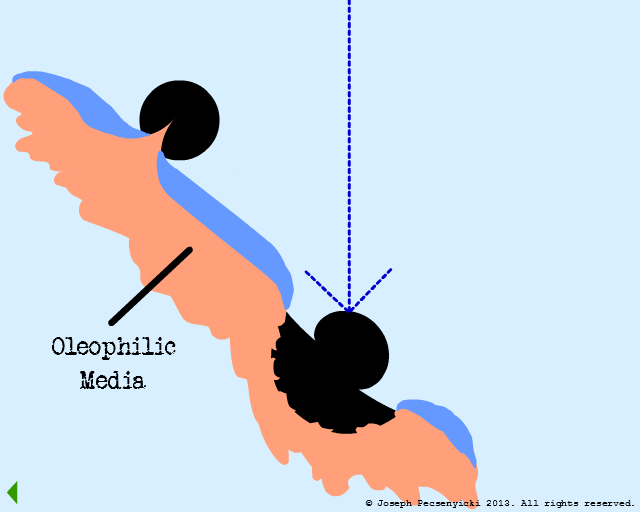
However, the asperities and pores of the walnut shell DO capture oil droplets (as per Model #1, Part III).
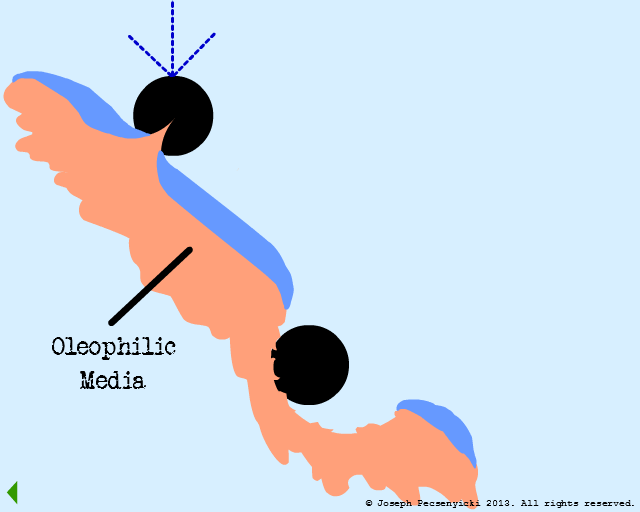
VI
Other droplets coalesce with the captured droplets.
VII
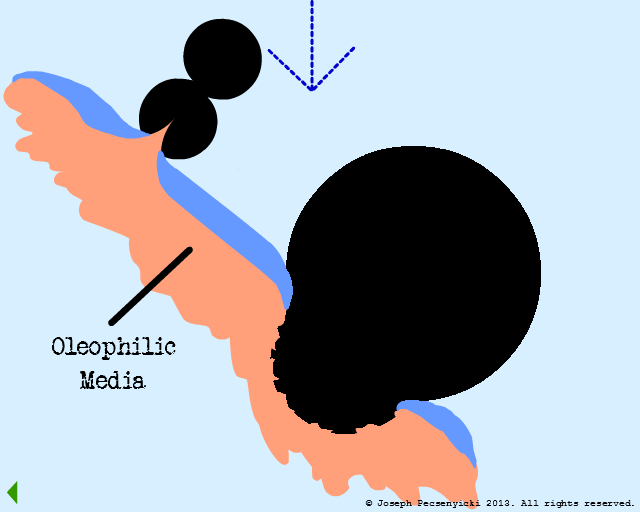
When the coalesced droplets within the pores reach a certain size, the surface tension breaks.
This wets the pores of the black walnut shell filter completely with oil (in a manner somewhat similar to Model #1, Part VIII).
VIII
The captured oil on the asperities and pores is strongly oleophilic, and serves as sites of capture for other droplets in the influent.
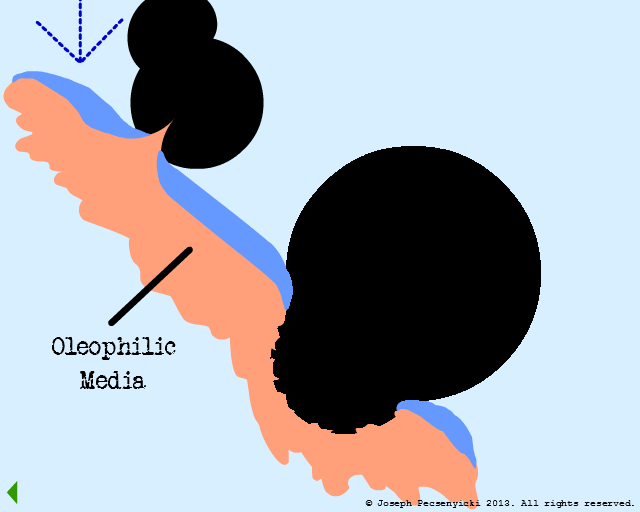
IX
The black walnut shell filter is now completely saturated with oil.
X
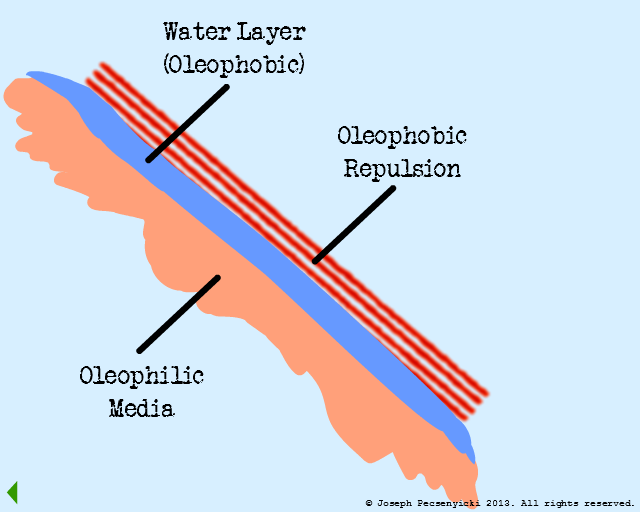
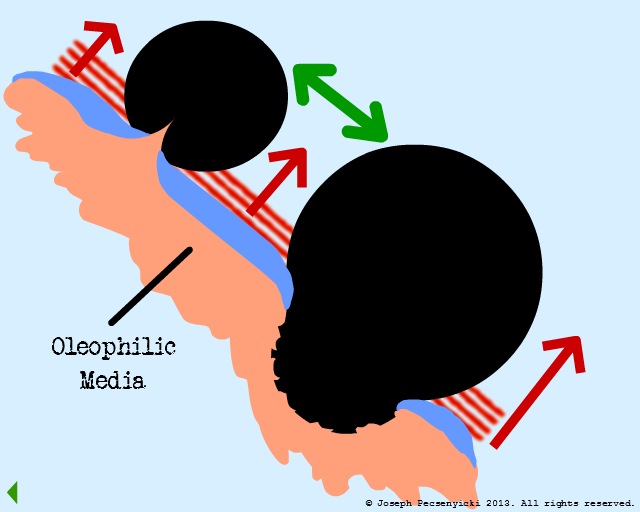
At this stage, the forces in the system are in equilibrium:
On the one hand, the oleophilic forces (green arrows) draw the captured oil droplets together.
On the other hand, the oleophobic forces from the water film (red arrows) serve to repel the oil just enough to keep it in droplet form. (Similar to rain drops beading on the surface of a newly-waxed automobile).
XI
However, if the system continues to operate, the filter will become fouled with oil.
This occurs because the captured oil droplets become too large and coalesce with each other.
The oil would then coat the surface irreversibly, as in Model #1, Part VIII.
XII
To prevent the filter from becoming fouled, it is periodically BACKWASHED.
This entails reversing the direction of the flow at high pressure, using relatively "clean" water as the influent.
XIII
During the backwash, the high-pressure influent loosens & removes captured oil droplets from the black walnut shell filter.
The large droplets are routed to a holding pond or slop tank where they coalesce further with each other.
XIV
The backwash has served its purpose, with the black walnut shells having been cleansed for the most part of captured oil droplets.
XV
Normal operation resumes, with the influent flowing once again from the top of the filter to the bottom.
The filter can now be re-used to capture more small oil droplets (Part VIII).
Credits
Static images from: Walnut Shell Filter Flash Animation
This animation was originally part of a more macro-level explanation given in a Chem 260 PowerPoint presentation.
The PowerPoint presentation itself was based in part upon a walnut shell filter presentation by Mike Howdeshell, Chris Catalanotto and Eric Lorgeused.
Header font: Veteran Typewriter by Magique Fonts
Paragraph font: My Underwood by Tension Type
Special thanks again to Ian Mackay and Kari McDonald.